Claria Preimplantation Genetic Testing
Preimplantation Genetic Testing- Aneuploidy (PGT-A)
Preimplantation Genetic Testing- Aneuploidy (PGT-A) is a test that examines the chromosomal material of an IVF embryo before implanation. It involves removing one or more cells from an IVF embryo to test for chromosome number. It checks for any numerical chromosomal abnormalities (Aneuploidy). This screening methods facilitates the selective implantation of Embryos with the normal number of chromosomes (Euploid).
1. One in Two human IVF embryos were observed to be chromosomally abnormal
2. Up to 40% of morphologically normal embryos are known to harbour chromosomal aneuploidies
3. PGS reduces number of IVF cycles required to get pregnant
4. PGS increases success rates for single embryo transfer
5. PGS Reduces the likelihood of miscarriage due to aneuploidies
1. Any women undergoing IVF
2. Women who have had repeated implantation failure or recurrent pregnancy loss while undergoing IVF
3. Women over 35 years of age undergoing IVF
4. Women with recurrent miscarriages after IVF
5. Positive history of chromosomal aneuploidies in the family
6. Diagnosed carriers of chromosomal aberrations
Preimplantation Genetic Testing- Monogenic (PGT-M) Structural Rearrangements (PGT-SR)
PGT-M, SR is a diagnostic procedure to test the material collected from an embryo for the presence of mutations carried by one or both parents. This is carried out when one or both genetic parents has a known genetic abnormality. In PGT-M, SR every test is prepared on a case-by-case basis.
1. Carriers of X-linked genetic disorders
2. Carriers of single gene disorders
3. Couples who have a child/ children who have are affected by a single gene disorder.
4. Couples who have a family history of a single gene disorder.
1. PGD can test for most single gene disorders.
2. The procedure is performed before implantation thus allowing the couple to decide if they wish to proceed further.
3. PGD can help select an embryo free from the single gene disorder affecting the parents, thus preventing future generations from being affected by the same disorder.
Why Trust Claria PGT-A, M, SR ?
With our wealth of technical expertise in Genomics, we are able to find the right solution for your needs.
Inherent flexibility to suit your needs without any compromise on quality.
Robust sequencing technology that provides sensitive and reproducible results.
We provide End to end support for the PGT-A, M, SR process: Right from site validation, embryo biopsy training to result data interpretation and phenotype correlation.
Free Genetic Counselling to guide you through the entire process
What are the advantages of NGS based technique ?
Medgenome Claria PGT is carried out using advanced Next Generation Sequencing technology.
- Rapid and Convenient
- Screening of all 23 pairs of chromosomes for abnormalities in one test
- Higher resolution-:Able to detect >20 Mb gains and losses in chromosome
- 1Mb areas are analysed to provide data with high confidence
- High Sensitivity in detecting aneuploidy (100 % sensitivity)
- High specificity and accuracy (99.98% specificity)
- Lower chances of test failure with NGS
Externally validated results
MedGenome’s PGT test has shown 100% accuracy in aneuploidy detection, when external validation was done using Proficiency Testing (PT) samples by the American Association of Bioanalyst
Superior to traditional screening methods
Fluorescent In Situ Hybridisation (FISH) |
Array Comparative Genomic Hybridisation (aCGH) |
Single Nucleotide Polymorphism (SNP) Microarray |
NGS |
|---|---|---|---|
| A largely manual process, highly skill/operator dependant | Requires Normal DNA for each sample to provide a comparison. A prolonged hybridisation step | SNP array analysis of DNA, extracted from a cell population, cannot indicate the mosaicism within the sample | NGS detects partial chromosomal gains and losses more precisely. NGS detects Aneuploidy and segmental imbalances at the same time |
| Screening all 24 chromosomes at once requires a special probes set, requiring separate software at higher cost per sample | Levels of mosaicism of 20% or less will not be detected | Longer time needed to complete intended test | NGS provides more accurate detection of mosaicism of the Trophectoderm cells from blastocyst biopsy |
| Difficult to resolve chromosomal overlaps/split signals | Relatively expensive | Relatively expensive | NGS offers reduced costs and enhanced precision. It allows parallel analysis for multiple embryos for a single patient. In a single run, samples from different patients can be analysed together as well |
What can Claria PGT detect ?
- Turner Syndrome
- Klinefelter Syndrome
- Down Syndrome
- Edwards Syndrome
- Patau Syndrome
- Other trisomies which may contribute to the risk of miscarriages
- Other monosomies which may contribute to embryo implantation failure
- Segmental Gain and Losses (>20Mb) in chromosomes which can lead to abnormalities in the embryo
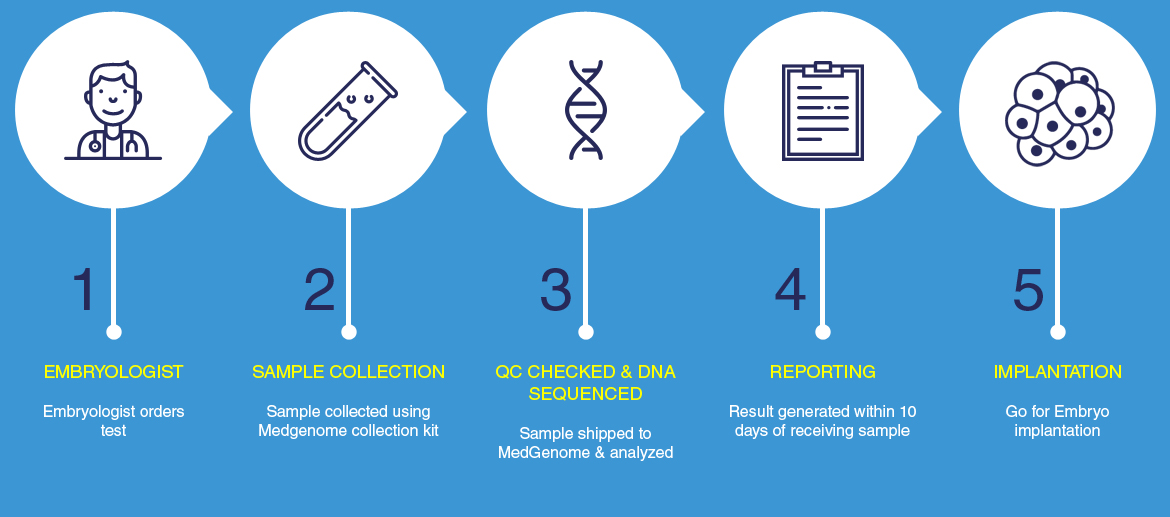
The Test Process
PGT Kit Ordering Procedure
Contact Customer Support at
MedGenome Labs Ltd. Bangalore
At least 2 Days in Advance
Clearly indicate the number of kits required
Kits will be transported to the provided
address at Room Temperature
Ensure the contents of the kit are intact before proceeding with the biopsy.
Refer to instruction on kit
